Clinical Trial Supply
Comparator sourcing, medicines and ancillaries
Drug developers preparing for clinical trials require support to ensure they have access to the necessary comparator and support medications, where and when patients need them.
We provide strategic insights and global comparator sourcing solutions to support your planning and comparator sourcing teams in their efforts to set up regulatory compliant, efficient, and value-adding clinical trials.
Benefits when outsourcing clinical trials:
- Strategic savings
- Agility and flexibility
- Trial-long, uninterrupted supply
- Reduced lead times and overstock
Strategic consultation
- Procurement Trends
- Emerging markets knowledge
- Cost and waste reduction
- Product Information
- Risk Analysis
Sourcing solutions
- Global Supply Network
- Reliable Supply
- Quality Compliant
- Documentation Offered
- Specific Lots Available
Why partnering with us for Clinical Trial Supply?
Our global logistics and distribution network for clinical manufacturing allows us to operate across borders and ensure supplies reach trial sites on time.
We are aligned with the needs of your company and the patients who depend on us working effectively together. Our global coverage and market leading support worldwide means one-stop control and visibility of your studies’ requirements.
Key Account Management and governance plans tailored to your needs ensure that your requirements will always be our priority.
One-Stop-Shop – Strategic Value for Clients
Sourcing – Storing – Packaging – Labelling – Distributing – Destructing
Key Strengths
- IMP and Co-Medication Supply Chain Solutions
- Strategic Consulting from Global Expert Team
- Dedicated Resources
- Optimized Sourcing, Manufacturing and Distribution Strategy
- Quick Quotations
- No More Waiting In Line!
- Site Ordering & Inventory Management Solution
- Central Pharmacy Model
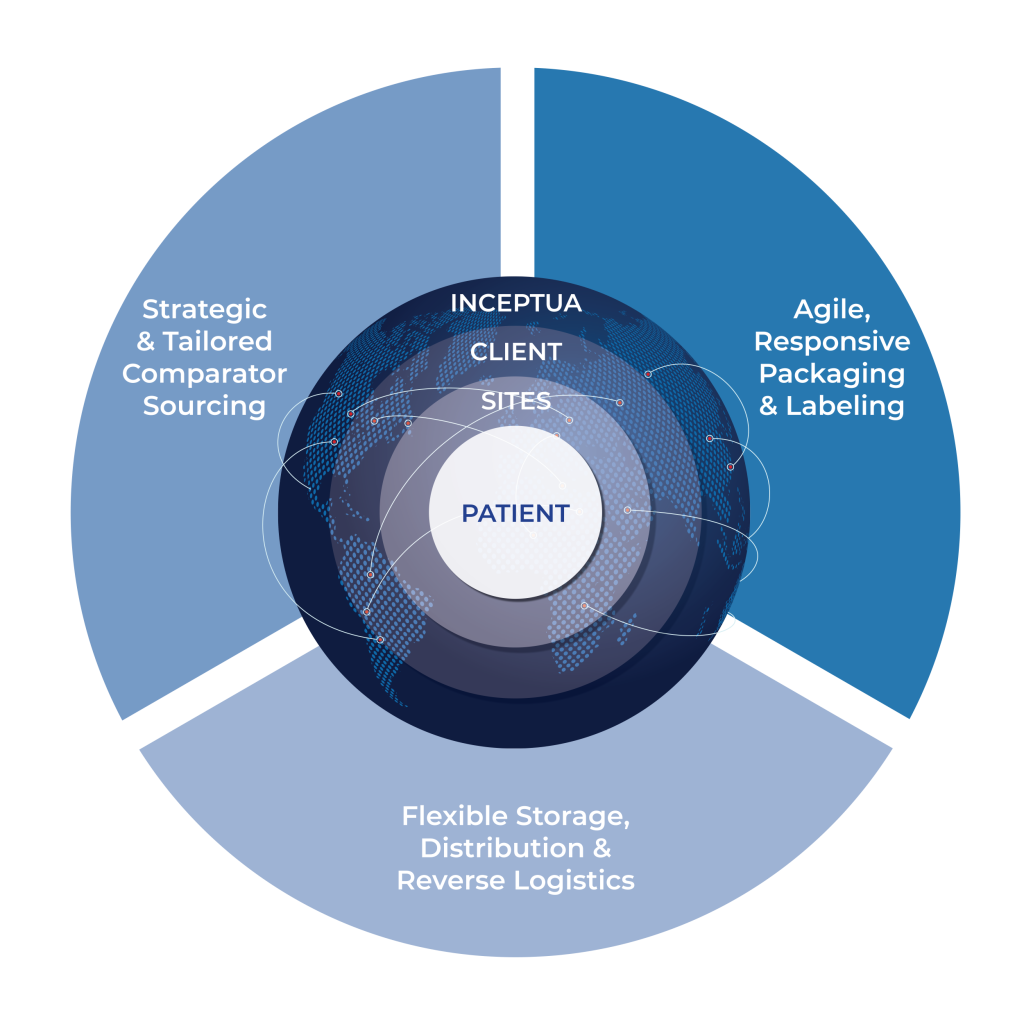
Client Benefits at a Glance
- Strategic cost savings and efficiency gains
- Reliable comparator sourcing worldwide
- Trial-long, uninterrupted supply chain
- Reduced lead times and minimized overstock
- Compliance with international regulatory standards
- A single, experienced partner managing end-to-end supply
Experienced, Transparent, Easy-to-Work with, providing Innovative, Agile & Responsive Solutions
We are experts in comparator sourcing
across major and rare therapeutic areas
In 2024, we delivered over 500 medicines, benefitting more than 100,000 patients across the world.
- Breast Cancer
- Colon Cancer
- Pancreatic Cancer
- Chronic Lymphocytic Leukemia (CCL)
- Hodgkin Lymphoma
- Prostate Cancer
- Wilms Tumor and other Pediatric Kidney Cancers
- Multiple Myeloma
- Testicular Cancer
- Chrohn’s Disease
- Colic
- High Cholesterol
- Blood Clots (DVT)
- Nutrition (parenteral nutrition PN)
- COVID-19
- Glucose, Saline, supplements and misc. diluents
- Fungal infections
- Thrombocytopeni
- Non-Small Cell Lung Cancer
- Myeloproliferative Neoplasms
- Acute Lymphoblastic Leukemia (ALL)
- Gastroesophageal Junction Cancer
- Kaposi Sarcoma
- Small Cell Lung Cancer
- RCC
- Meningeal Leukemia
- Cervical Cancer
- Ulcerative Colitis
- Plaque Psoriasis
- Myocardial Infarction
- Nausea
- Diabetes
- Asthma
- Tuberculosis (TB)
- Ancillaries
- Inflammation
- Other Vaccines
- Ovarian, Fallopian, or Primary Peritoneal Cancer
- Rectal Cancer
- Bladder Cancer
- Head and Neck Cancers
- Neuroblastoma / Globlastoma
- Soft Tissue Sarcoma
- HCC
- Mantle Cell Lymphoma
- Chronic Myeloid Leukemia
- Psoriatic Arthritis
- Pain indications
- Acute Transplant Rejection
- Cardiovascular Disease
- Gestational Trophoblastic Neoplasia
- Alzheimers Disease
- Schizophrenia
- Seizures
- COPD
- Acute Myeloid Leukemia (AML)
- Stomach (Gastric) Cancer
- Bone Cancer
- Bowel Cancer
- Non-Hodgkin Lymphoma
- Thyroid Cancer
- Metastatic Melanoma
- Myelofibrosis
- Arthritis
- Age-related Macular Degeneration (AMD)
- Anemia
- Macular (Dry Eye, AMD, RVO, DME, DR)
- Blood Cancer
- Brain Tumors
- Influenza
- Bacterial Infections
- Idiopathic Pulmonary Fibrosis (IFP)
Looking for packaging, labelling, storage and distribution services?
Fully compliant with local and international regulatory requirements
View moreAny questions?
Contact our experts